Computing drug-target binding kinetics: three papers in the Journal of Chemical Theory and Computation
How long does a drug molecule stay bound to its protein target? How quickly does it bind to and dissociate from its protein target? The MCM group works on developing computational methods to answer these questions. In three papers that have recently appeared in the American Chemical Society Journal of Chemical Theory and Computation, the researchers describe several computational approaches to study drug-target binding kinetics and provide insights into their determinants. These methods can help to guide the design of compounds with improved binding kinetics and thus better in vivo efficacy. The projects were funded by several programs of the European Union and the Klaus Tschira Foundation.
Daria Kokh and Rebecca Wade showed how the tRAMD method, which has been developed at HITS, can be applied to G protein-coupled receptors, important drug targets that are transmembrane proteins. This computationally efficient method reproduced relative residence times and revealed the dissociation mechanisms and the effects of allosteric modulation for two G protein-coupled receptors.
In a collaboration with Marc Bianciotto and colleagues at Sanofi R&D (France), the scaled molecular dynamics and tRAMD methods were assessed for the estimation of residence times and new techniques for the analysis of simulated trajectories were introduced. The results indicate how these approaches could be used for lead optimization or virtual screening in a drug design project.
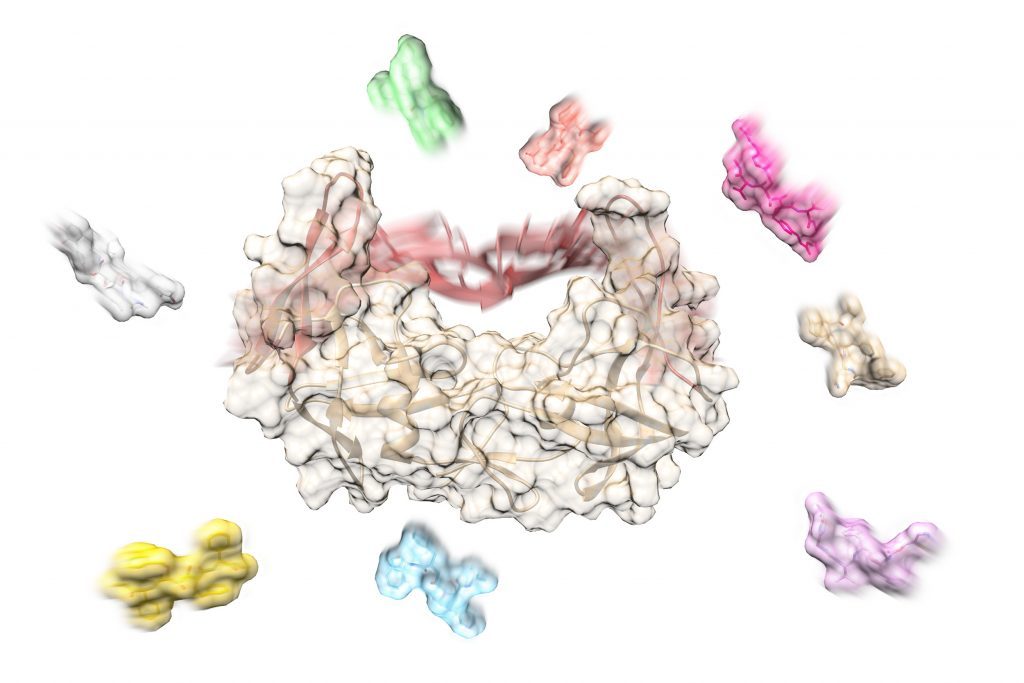
Kashif Sadiq et al. developed a multiscale approach to compute association rates for drug-like compounds that bind to a gated protein binding site. The first step is to perform molecular dynamics simulations of the unbound protein and to analyze the trajectories to identify the main conformations adopted, their population and ligand accessibility, and their interchange kinetics. In the next step, Brownian dynamics simulations of the diffusion of the molecules to the open states of the protein are performed to compute association rates, that are then adjusted by the gating factor computed for the unbound protein. Application of this approach to the binding of inhibitors to HIV-1 protease showed that conformational gating only modestly slows inhibitor binding despite the presence of two flaps that close over the binding site (see figure) and that other factors may limit association rates. For systems modulated by conformational gating, the approach can be scaled computationally efficiently to screen association kinetics for many ligands.
Funding: EU/EFPIA Innovative Medicines Initiative (IMI) Joint Undertaking, K4DD (Grant No. 115366), European Union Horizon 2020 Framework Programme for Research and Innovation under the Specific Grant Agreement No. 720270, 785907 and 945539 (Human Brain Project SGA1, SGA2 and SGA3), Klaus Tschira Foundation…
Publications:
Sadiq SK, Muniz Chicharro A, Friedrich P, Wade RC (2021). Multiscale Approach for Computing Gated Ligand Binding from Molecular Dynamics and Brownian Dynamics Simulations, J. Chem. Theory Comput., 17 (12), 7912–7929
Bianciotto M, Gkeka P, Kokh DB, Wade RC, Minoux H (2021). Contact Map Fingerprints of Protein–Ligand Unbinding Trajectories Reveal Mechanisms Determining Residence Times Computed from Scaled Molecular Dynamics, J. Chem. Theory Comput. 17(10):6522-6535
Kokh DB, Wade RC (2021). G Protein-Coupled Receptor–Ligand Dissociation Rates and Mechanisms from τRAMD Simulations, J. Chem. Theory Comput. 17(10):6610-6623
About HITS
HITS, the Heidelberg Institute for Theoretical Studies, was established in 2010 by physicist and SAP co-founder Klaus Tschira (1940-2015) and the Klaus Tschira Foundation as a private, non-profit research institute. HITS conducts basic research in the natural, mathematical, and computer sciences. Major research directions include complex simulations across scales, making sense of data, and enabling science via computational research. Application areas range from molecular biology to astrophysics. An essential characteristic of the Institute is interdisciplinarity, implemented in numerous cross-group and cross-disciplinary projects. The base funding of HITS is provided by the Klaus Tschira Foundation.